

Alpha decay causes the isotopes to lose 2 protons and 2 neutrons. Alpha particle bears a +2 electrical charge because there are no electrons to neutralize the positive charges of 2 protons. It is composed of 2 protons and 2 neutrons. The radioactive decay of substances may occur in three major ways.Īn alpha particle is identical to a Helium atom. If the ratio is too low, then it is proton rich atom and is unstable. If this ratio is too high, it is neutron rich and is unstable. Other atoms also can undergo radioactive decay according to their neutron: proton ratio. Therefore, these atoms can undergo radioactive decay. If there are too many protons and too many neutrons, the atoms are heavy. These nuclei undergo radioactive decay in order to become stable. An atom can become unstable due to several reasons such as the presence of a high number of protons in the nuclei or a high number of neutrons in the nuclei. Unstable isotopes are atoms having unstable nuclei. Radioactive decay is the process in which unstable isotopes undergo decay through emitting radiation. Key Terms: Half Life, Isotopes, Neutrons, Protons, Radioactive Decay What is the Relationship Between Radioactive Decay and Half Life This is the relationship between radioactive decay and half life. The time taken by a substance to become half of its initial mass through radioactive decay is measured as the half life of that substance. The radioactive decay of a certain substance is measured by a special term known as the half life. However, the final product of radioactive decay is always stable than the initial isotope. The radioactive decay causes an isotope of a particular element to be converted into an isotope of a different element. Therefore, in order to become stable, these isotopes undergo a spontaneous process called radioactive decay. Then the material is only slightly radioactive, and is sometimes treated as not harmful to people.There are certain naturally occurring isotopes that are unstable due to the imbalanced numbers of protons and neutrons they have in their nucleus of atoms. Carbon-14, which is used to find the age of fossils, has a half-life of 5,730 years.Īfter ten half-lives, over 99.9% of the radioactivity is gone. Plutonium-238 has a half-life of 88 years. As an example of fission decay, a fermium-256 atom can split into xenon-140 and palladium-112 atoms, releasing four neutrons in the process.įor example, uranium-232 has a half-life of about 69 years. For example, a radioactive carbon-14 atom releases a beta particle to become nitrogen-14. This makes them shake until they begin to decay and change into completely different types of atoms by releasing certain types of radioactive particles, such as alpha particles, beta particles or gamma rays, and possibly by fissioning into smaller nuclei. Their nuclei are unstable because the arrangement of protons and neutrons in them are unsteady. But if this experiment is repeated again and again, it will be seen that the atom decays within the half life 50% of the time. Taking one atom of 14C, this will either have decayed after 5,730 years, or it will not. Carbon-14 has a half-life of 5,730 years. Half-life is the expected value when half the number of atoms have decayed.
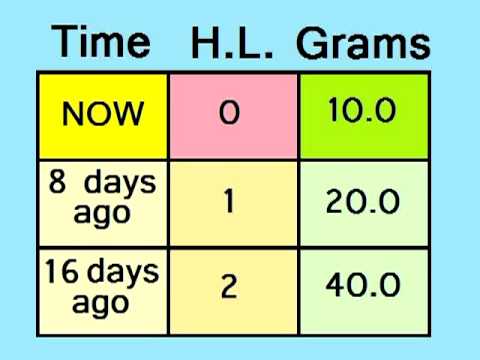
Note that half-life is defined as a probability. A Geiger-Muller detector can be used to measure the half-life it is the time when the activity is half of the original. It is now used in many other situations where something declines exponentially. The word "half-life" was first used when talking about radioactive elements where the number of atoms get smaller over time. The half-life of a substance is the time it takes for half of the substance to decay. Note the law of large numbers: With more atoms, the overall decay is more regular and more predictable. The number at the top is how many half-lives have elapsed. Simulation of many identical atoms undergoing radioactive decay, starting with either 4 atoms per box (left) or 400 (right).
